


In our first Blog Post (Part 1/3) of the series, we talked about the structure of the intestinal barrier and consequences of increased intestinal permeability. The intestinal barrier can be divided into three levels of protection: 1) gut flora, 2) intestinal mucosa, and 3) gut-associated immune system. If this barrier is compromised, increased permeability of the epithelium may occur. Transport through the epithelium is a highly regulated process. In this context, a distinction is made between paracellular and transcellular transport. While transcellular transport is an active transport of mostly small molecules through the epithelial cells, paracellular transport, on the other hand, is a passive diffusion of larger molecules between the epithelial cells. Tight junctions play an important role in the regulation of paracellular transport. Tight junctions are protein clusters that form on epithelial cell membranes and interact with adjacent epithelial cells. The interaction of two adjacent epithelial cells creates a diffusion barrier. This reversible interaction allows regulated substance diffusion through tight junctions.
Lactulose Mannitol Test – Gold Standard with Limitations
The gold standard in the diagnosis of increased intestinal permeability is the lactulose-mannitol test. The principle of the test is based on the measurement of two water-soluble sugars in the urine after their enteral absorption. The smaller sugar molecule mannitol is absorbed transcellularly, whereas the larger disaccharide lactulose is absorbed paracellularly. Due to the presence of tight junctions, only minimal amounts of lactulose are absorbed under physiological conditions. If the permeability is increased in the intestine, lactulose concentrations in urine increase and the ratio between lactulose and mannitol shifts. However, this method has some limitations. For example, it is not entirely scientifically clear whether the two sugars use different transport pathways. Other problems include the lack of generally accepted reference values and a lack of standardized performance in laboratories.
In addition to the lactulose-mannitol test, other diagnostic options have become established in the context of increased intestinal permeability.
Zonulin – the sensitive biomarker to detect increased intestinal permeability
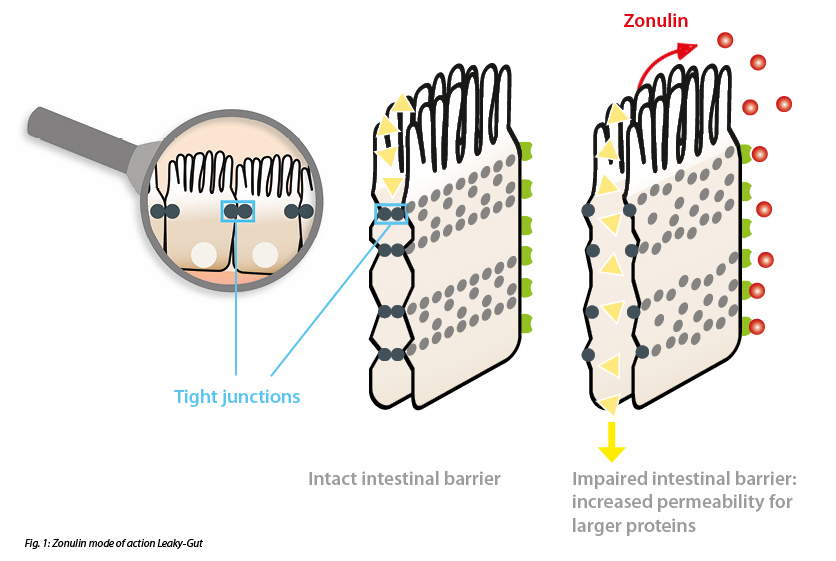
A widely used biomarker is zonulin. Zonulin is a human protein similar to the zonula ocludens toxin of vibrio cholerae that is involved in the regulation of tight junctions in the intestinal membrane. Zonulin binds to specific receptors on the surface of the epithelial cells of the intestinal barrier and activates a cascade of biochemical events that induce the opening of the tight junctions and, as a consequence, increase the permeability of the intestinal epithelial cells so that various substances can pass through the intestinal barrier and trigger autoimmune reactions. The readings of the IDK® Zonulin ELISA (K 5600 Stool) or IDK® Zonulin ELISA (K 5601 Serum), which detect zonulin family peptides, correlate with established metabolic traits related to increased intestinal permeability such as insulin resistance and obesity, as already established in many publications. (Fasano, 2011) (Mulak et al., 2019) (Caviglia et al., 2019)
α1-Antitrypsin – Permeability and Inflammation Marker
α1-Antitrypsin represents an inhibitor of inflammatory enzymes (elastases). As such an inhibitor, α1-antitrypsin is released during inflammatory processes to limit the proteolytic activity of elastases in the inflammatory area. α1-Antitrypsin is mainly synthesized in the liver or by intestinal macrophages, monocytes, and intestinal epithelial cells. Its fecal detection is considered a marker of intestinal protein loss and increased mucosal permeability, as it undergoes little intestinal degradation due to its antiproteolytic activity and is excreted almost unchanged in the stool. In addition, the determination of fecal α1-antitrypsin concentration is used to assess the activity of inflammatory bowel disease. In addition to the measurement of simple 24h-α1-antitrypsin excretion in stool samples, the α1-antitrypsin clearance determination has also become established in clinical routine (IDK® α1-Antitrypsin Clearance K 6752 – quotient of the α1-antitrypsin ELISA values of stool and serum samples). The IDK® α1-Antitrypsin ELISA (K 6750) is more sensitive than other commonly used detection methods and detects not only hepatic but also enteric α1-antitrypsin forms. Our ELISA thus represents a promising alternative for broad routine use. The combination of two specific antibodies in our IDK® α1-antitrypsin ELISA largely excludes the possibility of false negative results and thus ensures reliable diagnostics. (Faust et al., 2001)
FABP2 – Marker for damaged epithelial layer
FABP (fatty acid binding proteins) are a group of lipophilic proteins in the cytoplasm with diverse tasks, such as the uptake and transport of long-chain fatty acids or cholesterol. FABP2 is synthesized exclusively in the cytoplasm of intestinal epithelial cells and is therefore considered a specific marker for this type of cells. In the event of any structural damage to the epithelial cells, which for example occurs in an advanced leaky gut, FABP2 is passively released into the bloodstream. Thus, FABP2 in blood samples can be used as a biomarker for intestinal epithelial layer damage (IDK® FABP2 ELISA KR6809). (Adriaanse et al., 2016)
In principle, the combination of all biomarkers presented here is recommended for the diagnosis of increased intestinal permeability. Zonulin in stool is the most sensitive variant in this case. However, if increased intestinal permeability is already very advanced, severe structural damage of the epithelial layer may occur. This may result in low zonulin concentrations being measured because zonulin is synthesized by the epithelial cells themselves and secreted into the intestine. Low zonulin concentrations with simultaneously elevated α1-antitrypsin and/or also elevated FABP2 concentrations therefore indicate very severe damage to the epithelium.
In our next post Part 3, we will talk about diagnostic options for the gut immunological barrier associated with Leaky Gut.
References:
・Adriaanse, M. P., Leffler, D. A., Kelly, C. P., Schuppan, D., Najarian, R. M., Goldsmith, J. D., Buurman, W. A., & Vreugdenhil, A. C. (2016). Serum I-FABP Detects Gluten Responsiveness in Adult Celiac Disease Patients on a Short-Term Gluten Challenge. Am J Gastroenterol, 111(7), 1014-1022. https://doi.org/10.1038/ajg.2016.162
・Caviglia, G. P., Dughera, F., Ribaldone, D. G., Rosso, C., Abate, M. L., Pellicano, R., Bresso, F., Smedile, A., Saracco, G. M., & Astegiano, M. (2019). Serum zonulin in patients with inflammatory bowel disease: a pilot study. Minerva Med, 110(2), 95-100. https://doi.org/10.23736/S0026-4806.18.05787-7
・Fasano, A. (2011). Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer. Physiol Rev, 91(1), 151-175. https://doi.org/10.1152/physrev.00003.2008
・Faust, D., Spirchez, Z., Armbruster, F. P., & Stein, J. (2001). Determination of alpha1-proteinase inhibitor by a new enzyme linked immunosorbant assay in feces, serum and an enterocyte-like cell line. Z Gastroenterol, 39(9), 769-774. https://doi.org/10.1055/s-2001-17194
・Mulak, A., Koszewicz, M., Panek-Jeziorna, M., Koziorowska-Gawron, E., & Budrewicz, S. (2019). Fecal Calprotectin as a Marker of the Gut Immune System Activation Is Elevated in Parkinson's Disease. Front Neurosci, 13, 992. https://doi.org/10.3389/fnins.2019.00992
・Vanuytsel, T., Tack, J., & Farre, R. (2021). The Role of Intestinal Permeability in Gastrointestinal Disorders and Current Methods of Evaluation. Front Nutr, 8, 717925. https://doi.org/10.3389/fnut.2021.717925****