


In our second blog post of the series, we reported on the diagnostic possibilities of the epithelial barrier. Here, the biomarkers zonulin, α1-antitrypsin and FABP2 play a particularly important role. Besides diagnostic possibilities of the epithelial barrier, some parameters reflecting the immunological barrier status of the intestine have been established in the context of leaky gut.
Secretory IgA is an immunoglobulin produced by plasma cells located in the lamina propria and plays a crucial role in the immune function of mucous membranes. sIgA serves as the first line of defense of mucous membranes against pathogenic germs, toxins and allergens. In healthy individuals, sIgA coats mucous membranes with a continuous protective layer and is therefore found in body secretions such as saliva, tears, nasal mucus, tracheobronchial mucus, colostrum and breast milk, gastrointestinal secretions and stool. sIgA consists of two IgA monomers linked by a J-chain (“j” for “joining”). In addition, sIgA has a so-called secretory component, also called SC chain. This enables transport through the epithelial cells and stabilizes sIgA. Due to this special structure, sIgA, unlike IgA, is not degraded by enzymes. Therefore, the determination of sIgA in stool is very well suited for diagnostics. The determination of sIgA in stool with the IDK® sIgA ELISA (K 8870, 1-point calibratioin K 8880) provides information about the degree of stimulation as well as the secretion performance of the plasma cells located in the lamina propria of the intestine and therefore allows conclusions to be drawn about the state of the immunological barrier of the intestinal mucosa. A deficiency of sIgA indicates a reduced activity of the intestinal immune system, whereas increased sIgA values indicate an increased activity and thus a local inflammation of the intestinal mucosa. (Brandtzaeg, 1981; Strugnell & Wijburg, 2010)
In the absence of an immunodeficiency, low sIgA levels are an indication of increased intestinal permeability due to a non-functional immunological barrier.
Another important component of immunological protection in the intestine are defensins, endogenous antimicrobial peptides. These molecules are small cationic peptides that are similar in structure and always form three intramolecular disulfide bridges, stabilizing their characteristic β-sheet structure. This structure imposes an amphiphilic charge distribution that allows the defensins to interact with the target cell membrane and thus cause pore formation in the outer cell membrane, lysis, and possibly death of the cell. Human β-defensins-1 (hBD-1), 2, 3, and 4 are expressed by various epithelial cells. The normal intestinal mucosa expresses hBD-1, whereas hBD-2 and hBD-3 are produced only during inflammation. If there is a decreased β-defensin expression in stool samples (β-Defensin 2 ELISA K 6500), there is an impaired (Ganz & Lehrer, 1994; Hill et al., 1991)
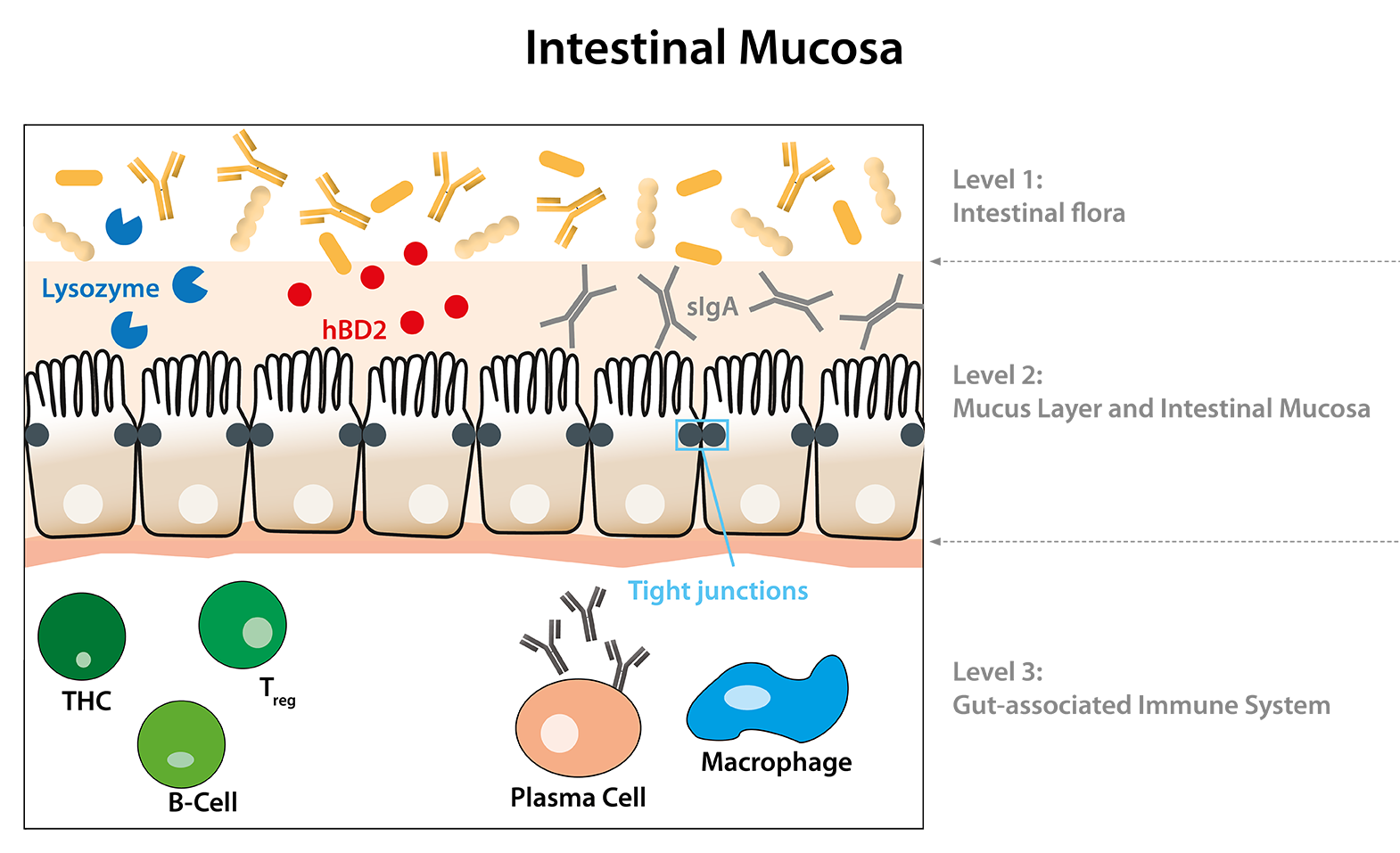
With the help of the broad product portfolio of Immundiagnostik AG in the area of leaky gut, statements can be made about both the epithelial and the immunological barrier of the intestine. For the epithelial barrier, primarily the biomarker Zonulin has been established. Increased zonulin concentrations lead to an active but also reversible opening of the tight junctions. Immundiagnostik AG offers zonulin ELISAs for stool as well as for serum (K 5600, K 5601). In contrast to zonulin, the biomarker α1-Antitrypsin is a passive marker for increased intestinal permeability. α1-Antitrypsin is a serum protein and diffuses through openings in the intestinal mucosa into the lumen of the intestine and can thus be detected in stool samples (K 6750, K 6760, K 6752). A combination of the biomarkers zonulin and α1-antitrypsin is recommended, as low zonulin concentrations can be caused by damage in the epithelium. This would lead to a false negative diagnosis. FABP2 is leaked by structurally damaged intestinal epithelial cells and can be detected in blood samples. Therefore, FABP2 is a biomarker for a severe impairment of intestinal permeability with epithelial damage (K 6809). Both sIgA (K 8870, K 8880) and hBD2 (K 6500) provide information about the immunological barrier of the intestine. If sIgA and/or hBD2 concentrations are decreased, the immunological barrier is not functional, which can lead to increased intestinal permeability.
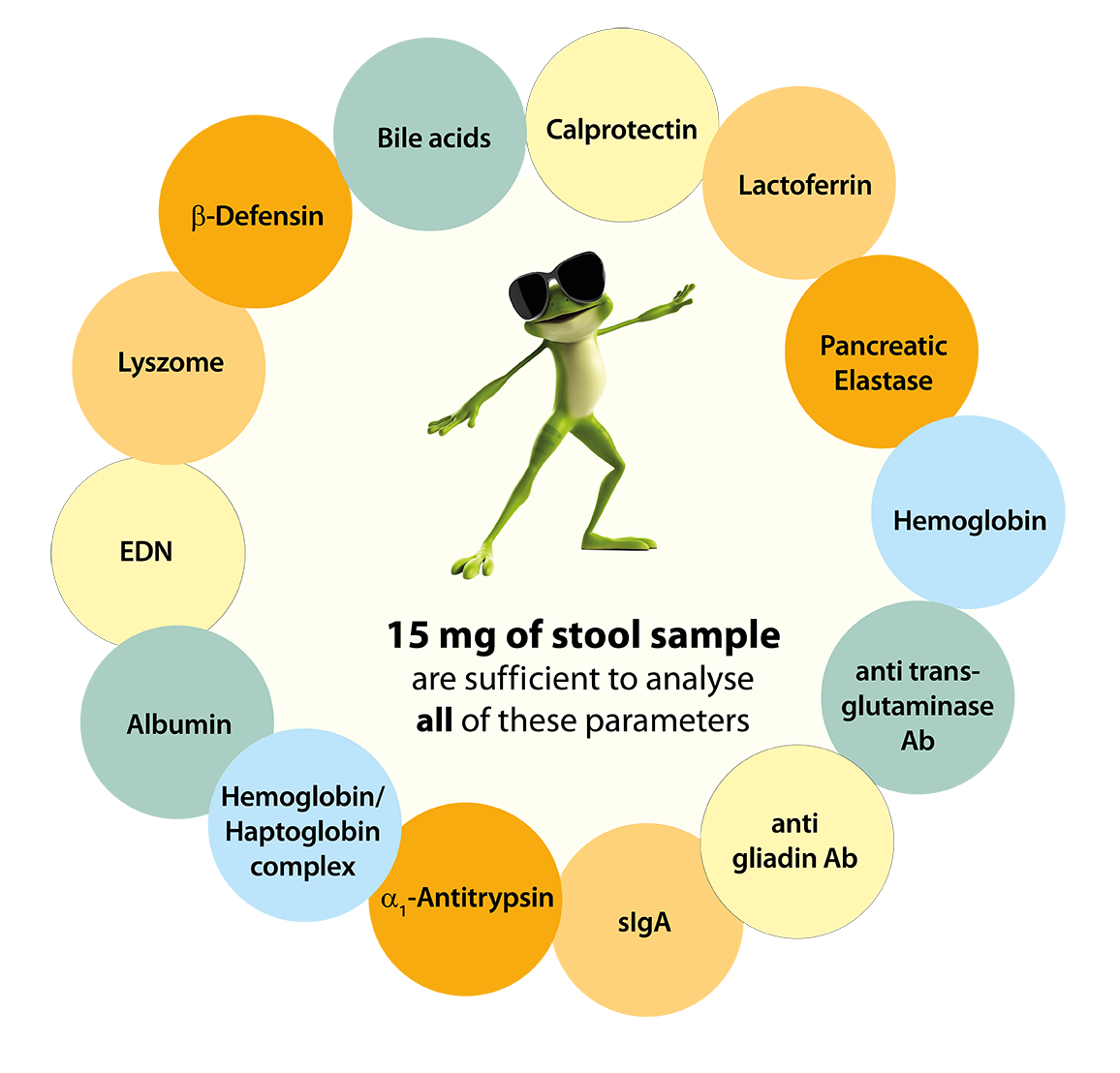
The parameters α1-antitrypsin, sIgA and hBD2 can be combined with the IDK® stool sample preparation tubes (SAS) filled with the extraction buffer IDK Extract® (K 6999). No sample weighing is required to obtain a defined sample quantity. When the yellow dip stick is inserted into the stool sample, the ridges at the tip of the stick fill with stool sample. When the dip stick is inserted into the stool sample tube, excess stool is wiped off, resulting in exactly 15 mg of stool entering the tube. The use of the IDK® SAS allows minimal stool contact and saves time and costs. The IDK Extract® extraction buffer included in the stool sample tube allows storage of the stool sample extract for 7 days at -20°C, 2-8°C and room temperature. The system is ready-to-use and can be used directly for manual as well as for automated processing.

References:
・ Brandtzaeg, P. (1981). Transport models for secretory IgA and secretory IgM. Clin Exp Immunol, 44(2), 221-232. https://www.ncbi.nlm.nih.gov/pubmed/6118214
・ Ganz, T., & Lehrer, R. I. (1994). Defensins. Curr Opin Immunol, 6(4), 584-589. https://doi.org/10.1016/0952-7915(94)90145-7
・ Hill, C. P., Yee, J., Selsted, M. E., & Eisenberg, D. (1991). Crystal structure of defensin HNP-3, an amphiphilic dimer: mechanisms of membrane permeabilization. Science, 251(5000), 1481-1485. https://doi.org/10.1126/science.2006422
・ Strugnell, R. A., & Wijburg, O. L. (2010). The role of secretory antibodies in infection immunity. Nat Rev Microbiol, 8(9), 656-667. https://doi.org/10.1038/nrmicro2384